This concept of the multiple bond preserves the idea of tetravalent carbon while admitting the existence of molecules in which carbon is clearly combined with fewer than four other atoms. You may be aware of the conventional way of depicting carbon as being tetravalent in all its compounds it is often stated that carbon always forms four bonds, but that sometimes, as in the case of ethylene, one of these may be a double bond. 
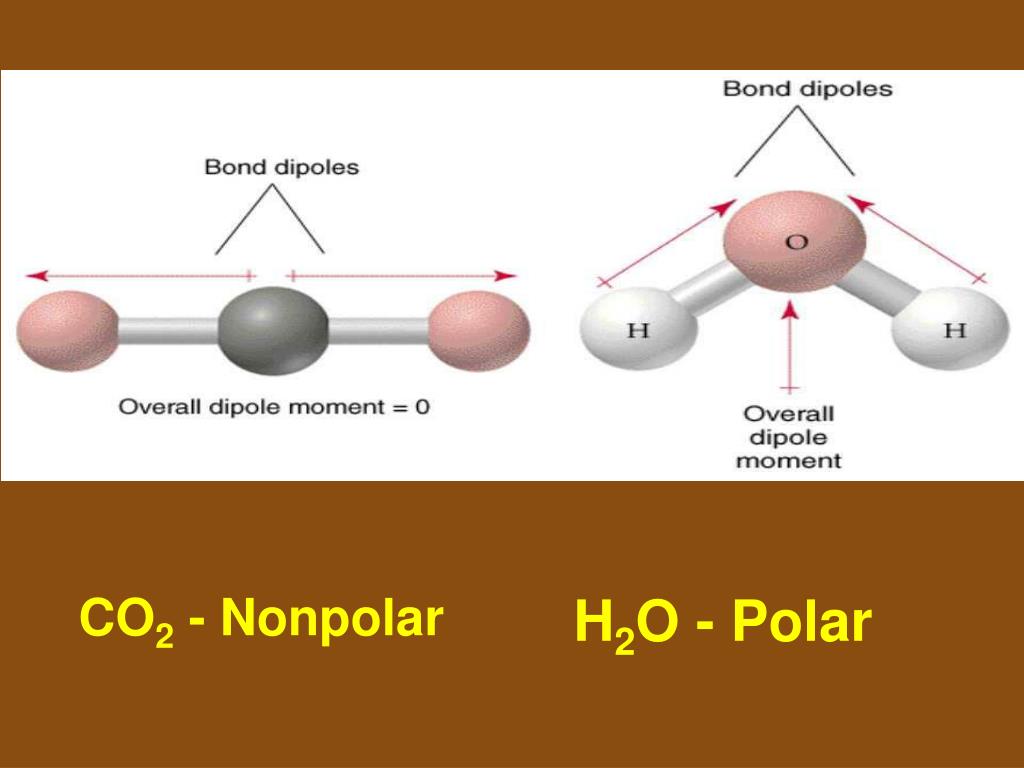
This alternative hybridization scheme explains how carbon can combine with four atoms in some of its compounds and with three other atoms in other compounds. Notice that the bond angles around each carbon are all 120°. Each carbon is bonded to three other atoms in the same kind of plane trigonal configuration that we saw in the case of boron trifluoride, where the same kind of hybridization occurs. We can explain this trivalence by supposing that the orbital hybridization in carbon is in this case not sp 3, but is sp 2 instead in other words, only two of the three porbitals of carbon mix with the 2 s orbital to form hybrids the remaining p-orbital, which we will call the i orbital, remains unhybridized. Here, we can regard carbon as being trivalent. Trigonal hybridization in carbon: the double bondĬarbon and hydrogen can also form a compound ethylene (ethene) in which each carbon atom is linked to only three other atoms. From the Lewis structure of CO 2 and VSEPR, we can determine that this is a linear molecule.\) Let's look at some of the physical properties of CO 2. The C-O bonds in carbon dioxide are polar and yet the dipole moment is zero because the 2 bond dipoles cancel each other. One thing that we can understand by looking at the structure of CO 2, is that the carbon center of the molecule must be electrophilic. An electrophile (electron-lover) is a center that is electron poor and will be attracted to centers that are electron-rich.Įven though the total electron count around the carbon is 8, this overestimates the electron density. This carbon is bonded only to highly electronegative oxygen atoms. The bonding electrons will all be more closely associated with oxygen than with carbon.įrom the Lewis structure we can see that the carbon in CO 2 must make 2 sigma bonds and it has no lone pairs. This atom will be 2sp hybridized with remaining 2p x and 2p y atomic orbitals.Įach oxygen makes 1 sigma bond and also needs 2 orbitals for lone pairs of electrons. These must each be 2sp 2 hybridized with a remaining 2p orbital. One of the oxygens will have a 2p x orbital to combine with the carbon 2p x orbital. The other oxygen will have a 2p y orbital that can combine with the other p orbital on carbon.Ī 2sp 2 orbital on O1 combines with a 2sp orbital on C to make a sigma bonding and a sigma antibonding molecular orbital. The other 2sp orbital on C combines with a 2sp 2 orbital on O 2 to make another set of sigma bonding and sigma antibonding molecular orbitals. The remaining 2sp 2 from the oxygen atoms become non-bonding molecular orbitals. The O 1 2p x combines with the C 2p x to make a pi bonding and pi antibonding molecular orbital. 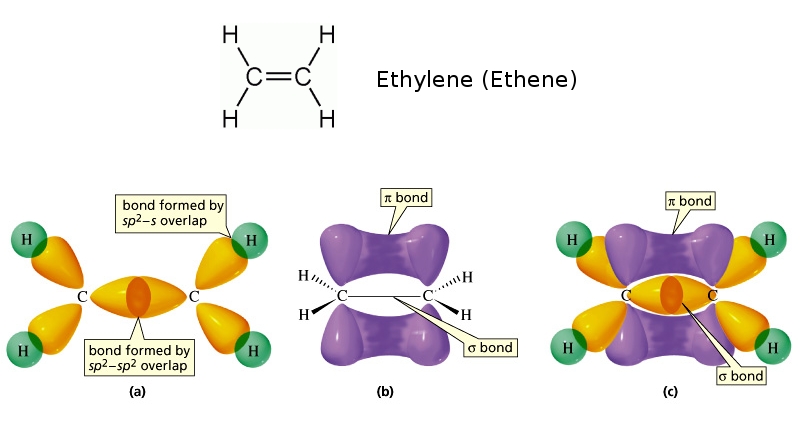
The O 2 2p y combines with the C 2p y to make another set of pi bonding and pi antibonding molecular orbitals. The 16 valence electrons fill through the 2 pi bonding orbitals so there is a full double bond between carbon and each oxygen.Īs you saw above, the total electron count around the atoms in carbon dioxide seriously overestimates the electron density around the carbon atom. It doesn't help us predict the reactivity of this atom. The oxidation state formalism can give us a better idea about the electron density around an atom and its tendency to add electrons and become reduced.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
